Introduction to Weather
The earth's atmosphere
Composition
The earth's atmosphere is composed of a few different gases, the percentage of each gas stays relatively the same with increasing altitude, but the amount of atmospheric pressure, temperature and air density decreases with increasing altitude.
In the earth's atmosphere the following gases are present:
- Nitrogen, 78 %
- Oxygen, 21 %
- Argon, ~1 % (0.93 %)
- Trace gases, 1 % (Carbon Dioxide, Neon, Helium, Methane, Krypton, Hydrogen, and Ozone)
This is the composition of a dry atmosphere here at earth, however it is rarely so, due to to water vapor. Water vapor can make up 0 to 4 % of the earth's atmosphere, and this will affect the other gases.
Vertical structure
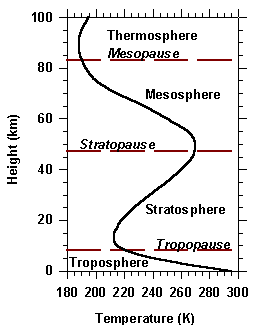
The vertical structure of the atmosphere can be divided into 5 different spheres, with each sphere being separated by a layer of stable temperature called a pause.
The Troposphere is the part of earth's atmosphere that is closes to the surface and extends up to 15 km. This layer contains approximately 75 % to 80 % of the earth's atmospheric mass and most weather phenomena occur here. With increasing altitude, the temperature decreases to around -56°C on average.
The Stratosphere is above the troposphere and they are separated by the tropopause. The stratosphere extends from around 15 km to 50 km. This layer contains the ozone layer, which absorbs UV radiation, thus increasing temperature with increasing altitude. Due to more stable conditions, most airliners fly in the lower stratosphere.
The Mesosphere is above the stratosphere and they are separated by the stratopause. The Mesosphere extends from around 50 km to 85 km. Here the temperature decreases again, and reaches its coldest point in the atmosphere which is about -90°C.
The Thermosphere is above the mesosphere and they are separated by the mesopause. The Thermosphere extends from 85 km to 600 km. This layer is characterized by extremely low density, with temperature increasing to 1500°C or more due to absorption of high-energy solar radiation.
Finally we have the Exosphere, which is the outermost edge, where atoms and molecules escape into space.
International Standard Atmosphere (ISA)
International Standard Atmosphere (ISA) is a term used to describe a set of internationally agreed upon standards which can be used to make basic calculations about temperature and pressure change. By following ISA flights happening at a high altitude can for example all use the same pressure settings so that they can be sure of vertical separation. There are limitations to this standard, as it is a dry, non-viscous model and does not include wind or turbulence. There are a few key things to remember:
- Temperature at sea level: 15°C
- Temperature lapse rate: -2°C (-1.98°C) per 1000 feet
- Pressure at sea level: 29.92 inHg (or 1013.25 hPA)
Temperature
Temperature will vary with altitude. If you have a parcel of air that contains the same volume of air, but one is warm and one is cold, their density would be different.
Two parcels of air with the same volume will have a higher density if the air is cold compared to it being warm.
This concept is useful when we talk about pressure systems and movement of air, so keep it in mind!
Atmospheric pressure and altimetry
Atmospheric pressure (or barometric pressure) is the force per unit area exerted by the weight of the air column above a location, caused by gravity acting on air molecules.
whiteboard or image of how this can be measured using a barometer with mercury for example.
Sea level pressure vs Station pressure awdawd
Pressure systems
High
Low

which direction in what hemisphere and movement of air
Wind
-Basic theory of general circulation
-Coriolis force
-Pressure gradient force (HIGH to LOW)
-Local wind systems (Land vs. Sea | Valley vs. Mountain)
-Friction effect (slows down wind, backs and veers)
Moisture
-Physical states (WHAT IS THIS MEANT TO BE ABOUT?)
Im thinking, state changes, gas to liquid condensation. etc etc
-Relative humidity, relative humidity calculation
RH = 100 - (temp - dew ) * 5 c
RH = 100 - (temp - dew ) * 25/9 f
explain what it is. 100% == saturated etc.
-Dewpoint
temp at which air must be cooled to become saturated and water vapor condenses into liqud.
dew point changes when the absolute amount of water vapor in the air changes or when atmospheric pressure changes.
Stability
ADIABATIC PROCESS, air chagnes temp without exchanging heat with surroundings, expansion and compression.
environmental laps rate
dry vs wet laps rate
-Dry adiabatic lapse rate
-Temperature and dew point convergence
Stable
Unstable
Neutral
drawing
Clouds,
-Formation and structure
- Condensation nuclei
- Water vapor
- Dew point met
-Types
height classification
-Recognition
looks
This lesson was last edited 1 month, 1 week ago.
Previous | Next
